 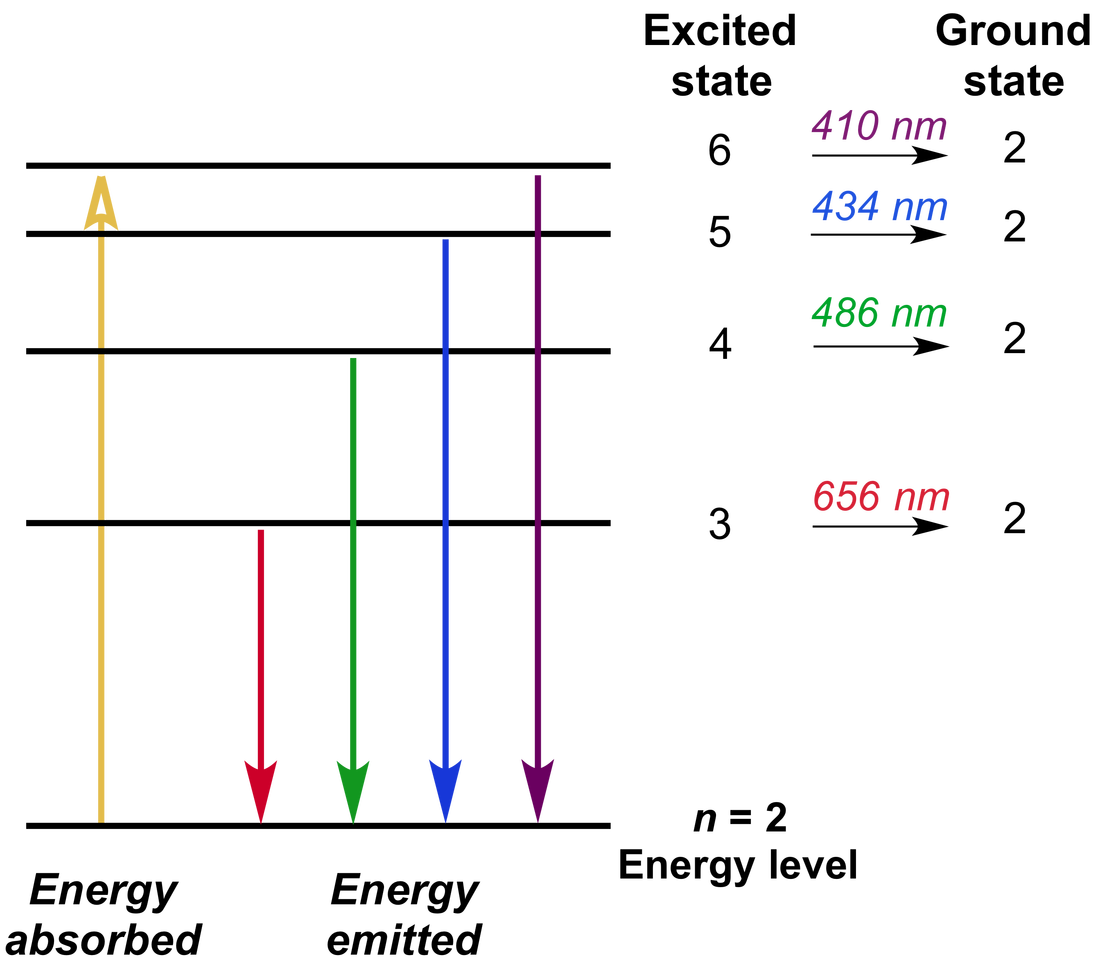
I think this is when white light is used that you get an Absorption Spectra. 
All the colors of the Absorption Spectra do make it kind of confusing. And these are being absorbed (with emphasis on blue). Actually, if you just burned hydrogen and looked at its spectra, you would get the Emission Spectra and not the Absorption Spectra, and this Emission Spectra would only show the bunch of blue lines, one purple line, and one red line. All the other colors shown are just part of the natural light being shown down on the element. Study with Quizlet and memorize flashcards containing terms like The lowest allowable energy state of an atom is called its, Bohrs model of the atom predicted the of the lines in hydrogens atomic emission spectrum., According to Bohrs atomic model, the smaller an electrons orbit, the the atoms energy level. When light from a hydrogen gas discharge tube is passed through a prism, the light is split into four visible. The Figure below shows the atomic emission spectrum of hydrogen. This is the color that will be the opposite of the flame color on the color wheel. An atomic emission spectrum is the pattern of lines formed when light passes through a prism to separate it into the different frequencies of light it contains. Remember, always look at the color area on the rainbow that is blacked out the most. So if blue is being absorbed, the opposite color would be transmitted and this color is orange. However, there are MORE dark lines in the blue region. Study with Quizlet and memorize flashcards containing terms like amplitude, atomic emission spectrum, Bohr model and more. If you look at the lines for hydrogen blue, purple, and red are being absorbed. Study with Quizlet and memorize flashcards containing terms like amplitude. Which of the following statements is true regarding these. Therefore, all the other colors would be absorbed. Atomic emission spectra are produced when the light emitted by an element passes through a prism. (This would be orange.) The element hydrogen turns orange when being burned and this color is transmitted to us. 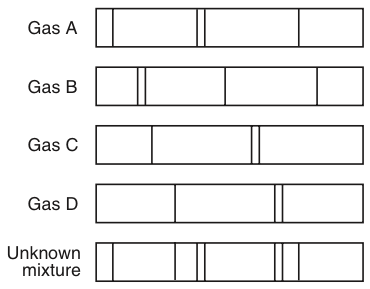
This means that if there is a big dark band where blue would be, then the opposite color to blue on the color wheel is being transmitted. electrons add E at ground state, jump to excited state, lose E and fall back down to emit E in form of light. Describe the cause of atomic emission spectrum of an element. You are supposed to look at the dark areas of the absorption spectra and those dark areas indicate that the color which would be there is being absorbed. each line in the spectrum corresponds to one exact frequency of light emitted by the atom. I think both the absorption and emission lines are showing which colors are being absorbed. Study with Quizlet and memorize flashcards containing terms like Which of these questions were left unanswered by Rutherfords atomic model, Light emitted by elements when heated revealed the arrangement of in atoms which explained the behavior of elements. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
