 
Combining the in-phase orbitals results in a bonding orbital. One contains the axis, and one contains the perpendicular. 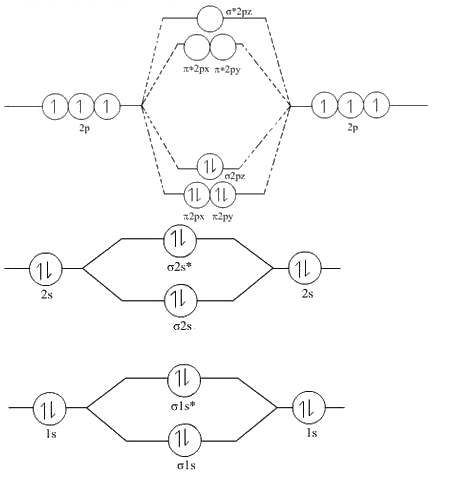
Combining the out-of-phase orbitals results in an antibonding molecular orbital with two nodes. Side-by-side overlap of each two p orbitals results in the formation of two π molecular orbitals. The molecular orbital configuration of CN is (1s 2 )( 1s 2 )(2s 2 )( 2s 2 )(2p x 2 )(2p y 2 )(2p 2 ). The bond order of CN is 3 while that of CN + is 2. For the out-of-phase combination, there are two nodal planes created, one along the internuclear axis and a perpendicular one between the nuclei.įigure 7.7.6. The molecular orbital diagram of CN shows the placement of electrons in molecular orbitals after C-N bond formation. Electrons in this orbital interact with both nuclei and help hold the two atoms together, making it a bonding orbital. In molecular orbital theory, we describe the \pi orbital by this same shape, and a \pi bond exists when this orbital contains electrons. In valence bond theory, we describe π bonds as containing a nodal plane containing the internuclear axis and perpendicular to the lobes of the p–\pi orbitals, with electron density on either side of the node. The side-by-side overlap of two p orbitals gives rise to a pi (\pi) bonding molecular orbital and a \pi* antibonding molecular orbital, as shown in Figure 7.7.6. I was just wondering if the same applied for molecules with a positive charge. and for molecules that have electrons from 15 to 20 have the order '1s 1s 2s 2s. Molecular Orbital Diagram of NO+ Postby Josh Ku 3H Tue 7:23 pm viewtopic.phpf43&t16787 In the link above chemmod said it was best to account for the negative charge of CN- by placing an extra electron on the nitrogen since it is more electronegative. Or, another way to write this is in the notation seen in the MO diagram, with KK indicating the filled 1s and 1s. For those that have less than or equal to 14 electrons we use the order '1s 1s 2s 2s 2px,2py 2pz 2px,2py 2pz'. So, the molecular electron configuration would be written in a similar manner as the atomic counterpart, but using molecular orbitals instead. Combining wave functions of two p atomic orbitals along the internuclear axis creates two molecular orbitals, σp and σ∗p. When we draw the molecular orbital diagram for molecules. Just as with s-orbital overlap, the asterisk indicates the orbital with a node between the nuclei, which is a higher-energy, antibonding orbital.įigure 7.7.5. There is an \ce^* (antibonding) (read as “sigma-p-x” and “sigma-p-x star,” respectively). This electronic structure adheres to all the rules governing Lewis theory. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
